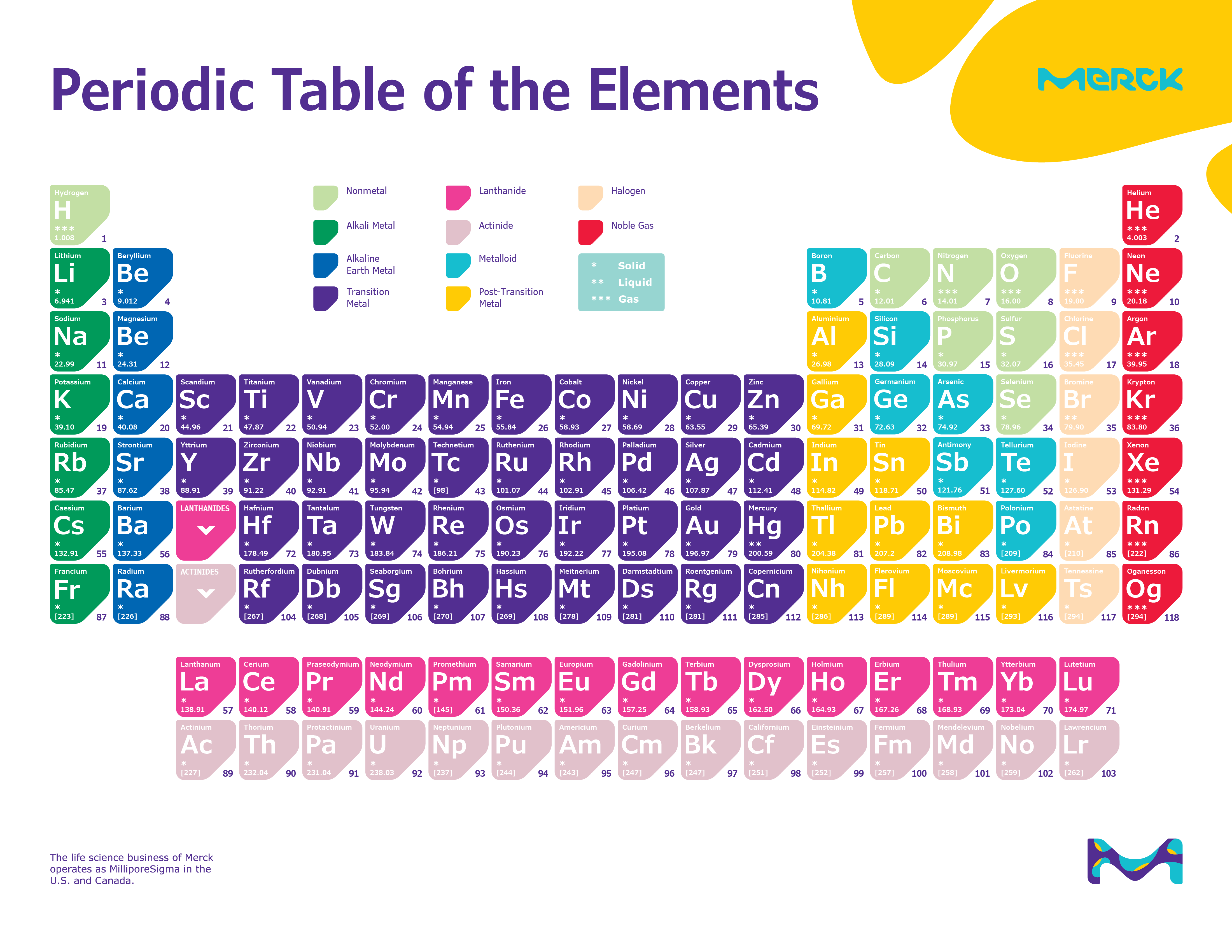
Students may be interested in the even smaller particles of quantum physics-like quarks, bosons, neutrinos, and antineutrinos. They are intended to get players thinking about what they already know about elements. General questions about the properties of elements assume standard temperature and pressure (helium is liquid below -268☌ and gold is a liquid above 1064 ☌). For this game, the most common isotopes of the chemical elements are used. This game presents a simple introduction to the Rutherford-Bohr model of the atom and the way we organize the elements. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. NS 5.8.2 - Develop an understanding of properties of matter Note: The Royal Society of Chemistry's interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Any bond or intermolecular attraction that can be formed can be broken.Chemical and physical properties of materials can be explained by the structure and arrangement of atoms, ions, and molecules and the forces between them.Matter is made from discrete fundamental units called atoms.Concepts:Ĭhemical elements, Periodic Table, chemical symbols, atom, atomic number, atomic weight, protons, electrons, neutrons, atomic structure. Interactive module that introduces atomic structure Learning objectives:Īfter this activity, the student will be able to describe the basic structure of matter, name the parts of an atom, have experience using the Periodic Table, explain elements, and have the background to understand isotopes. Teacher Information: Activity Description:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
